Have you ever wondered how a rare obstetric emergency like Amniotic Fluid Embolism Pathophysiology can turn a healthy childbirth into a life-threatening crisis within minutes? This mysterious condition leaves doctors and researchers racing against time to understand its underlying mechanisms. What exactly happens when amniotic fluid enters the maternal bloodstream, and why does it trigger such a catastrophic response?
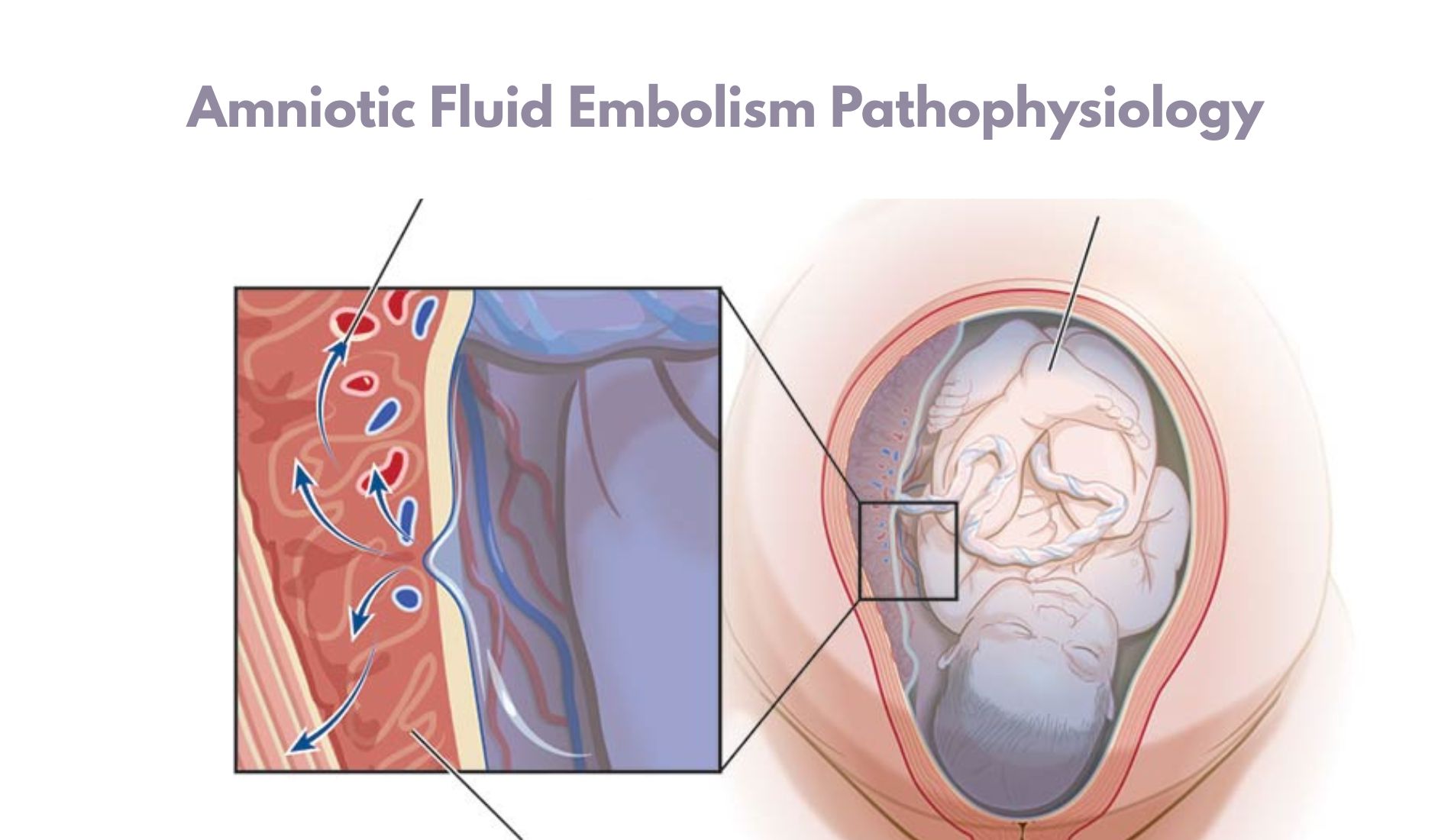
Amniotic Fluid Embolism (AFE) Pathophysiology involves a complex immune and cardiovascular reaction when fetal materials, such as amniotic fluid or debris, breach the maternal circulation. This leads to sudden respiratory distress, cardiac collapse, and coagulopathy. Experts like Dr. Steven L. Clark, a renowned obstetrician and AFE researcher, describe it as an “anaphylactoid syndrome of pregnancy,” emphasizing immune-driven shock rather than a true embolism.
But there’s much more to uncover about how this rare condition unfolds at the molecular and systemic level. Curious to know the latest insights and clinical findings shaping modern obstetric care? Let’s dive deeper into the science and unravel the fascinating yet terrifying world of Amniotic Fluid Embolism Pathophysiology.
What Is the Pathophysiology of Amniotic Fluid Embolism?
The pathophysiology of amniotic fluid embolism is multifactorial and not yet completely understood. Traditionally, it was thought to result from the physical entry of amniotic fluid, fetal cells, or debris into the maternal circulation, leading to mechanical obstruction of pulmonary vessels. However, recent studies suggest that the condition is more accurately described as an anaphylactoid reaction rather than a true embolic event.
In essence, when amniotic fluid or fetal antigens enter the maternal bloodstream—usually through uterine veins during labor, delivery, cesarean section, or uterine trauma—they trigger a massive inflammatory and immune-mediated response.
This cascade leads to pulmonary vasospasm, right ventricular failure, hypoxia, and ultimately cardiovascular collapse. The release of vasoactive substances such as prostaglandins, leukotrienes, and endothelins exacerbates pulmonary hypertension and reduces cardiac output.
Thus, AFE represents a two-phase pathophysiological process: an initial cardiopulmonary collapse followed by a hemorrhagic and coagulopathic phase.
Amniotic Fluid Embolism Pathophysiology Overview:
In modern medical understanding, Amniotic Fluid Embolism Pathophysiology involves a complex interplay of hemodynamic, immunologic, and hematologic disturbances. The sequence of events can be summarized as follows:
- Breach of the maternal-fetal barrier: During labor or delivery, small tears in the placental or uterine vessels allow amniotic fluid to enter the maternal venous system.
- Activation of the immune system: Once fetal antigens and debris reach the maternal lungs, the immune system recognizes them as foreign. This triggers a cascade similar to anaphylaxis, releasing histamine, complement factors, and cytokines.
- Pulmonary vasoconstriction and right heart failure: The sudden rise in pulmonary artery pressure causes acute right ventricular strain and decreased left ventricular preload. The patient experiences hypotension, hypoxia, and cardiovascular collapse.
- Disseminated intravascular coagulation (DIC): The exposure of tissue factor and activation of the coagulation pathway leads to widespread clotting followed by consumption of clotting factors and platelets. This results in uncontrolled bleeding.
The clinical course typically progresses rapidly, emphasizing the need for immediate recognition and supportive intervention.
What Are the Two Phases of Amniotic Fluid Embolism?
Amniotic Fluid Embolism unfolds in two distinct phases, each with characteristic clinical and physiological manifestations:
Phase 1: Cardiopulmonary Collapse
This is the initial phase, occurring within minutes of amniotic fluid entering the maternal circulation. The sudden onset of symptoms includes:
- Shortness of breath and chest pain
- Cyanosis
- Hypotension or cardiac arrest
- Pulmonary hypertension due to vasospasm
The primary pathophysiologic event here is acute right ventricular failure secondary to increased pulmonary vascular resistance. Oxygen delivery to vital organs plummets, leading to hypoxia, arrhythmias, and loss of consciousness.
Phase 2: Hemorrhagic Phase (Coagulopathy)
Once the patient survives the initial cardiopulmonary collapse, the second phase manifests as massive bleeding due to Disseminated Intravascular Coagulation (DIC). The coagulation cascade is activated systemically, leading to microvascular thrombosis followed by depletion of clotting factors. Clinical signs include:
- Uncontrolled postpartum hemorrhage (PPH)
- Uterine atony
- Oozing from intravenous or surgical sites
- Shock and multi-organ failure
The rapid transition between these phases underscores the lethal nature of AFE and the need for early diagnosis and aggressive management.
Diagnosis of Amniotic Fluid Embolism
Diagnosing AFE remains a clinical challenge due to its abrupt onset and nonspecific presentation. There is no single laboratory test that confirms the diagnosis. Instead, it is based on clinical suspicion and the exclusion of other causes such as pulmonary embolism, anaphylaxis, or eclampsia.
The Clark criteria (developed by the Society for Maternal-Fetal Medicine) are widely accepted for research and clinical categorization. These include:
- Sudden cardiovascular collapse or cardiac arrest.
- Acute hypotension or respiratory distress during labor, delivery, or within 30 minutes postpartum.
- Disseminated intravascular coagulation (DIC) following the initial event.
- Absence of any other explanation for the clinical presentation.
Supporting investigations may include:
- Arterial blood gases (showing hypoxemia and metabolic acidosis)
- Elevated pulmonary artery pressure (via echocardiography)
- Coagulation studies confirming DIC
- Chest imaging (revealing pulmonary edema or infiltrates)
While fetal squamous cells or debris may be detected in maternal pulmonary circulation, their presence is not diagnostic, as they can appear in asymptomatic deliveries as well.
Why Autopsies Are Essential in AFE Diagnosis? (Clinical Presentation)
Autopsies play a crucial role in confirming AFE, particularly in fatal cases where ante-mortem diagnosis is uncertain. The clinical presentation is often dramatic and abrupt, leaving little time for diagnostic evaluation during life.
Clinical Presentation:
Typical features of AFE include:
- Sudden respiratory distress, cyanosis, and collapse
- Profound hypotension or cardiac arrest
- Coagulopathy leading to massive bleeding
- Seizures or altered mental status
- Fetal distress if the event occurs before delivery
At autopsy, microscopic examination often reveals fetal squamous cells, lanugo hair, or mucin within the maternal pulmonary arterioles. However, the absence of these findings does not exclude AFE, as the reaction may be immunologic rather than mechanical.
Thus, autopsies not only confirm the diagnosis but also provide valuable insights into the mechanisms of AFE, contributing to medical research, quality improvement, and accurate mortality statistics.
What Happens During an Amniotic Fluid Embolism?
During an AFE event, the maternal immune and cardiovascular systems are overwhelmed by an acute insult. Here’s a step-by-step breakdown of what happens:
- Entry of amniotic material: Amniotic fluid containing fetal cells, hair, vernix, and trophoblastic tissue gains access to maternal circulation.
- Pulmonary vascular obstruction and vasospasm: These materials and associated mediators cause an immediate increase in pulmonary artery pressure.
- Right heart strain: The right ventricle fails to pump against elevated resistance, leading to decreased left ventricular output and systemic hypotension.
- Hypoxemia and tissue hypoxia: Gas exchange is impaired, resulting in severe hypoxia and cyanosis.
- Coagulation cascade activation: Tissue factor from the amniotic fluid triggers DIC, causing widespread clotting and bleeding.
- Cardiac arrest and shock: Without immediate resuscitation, multi-organ failure ensues, often culminating in maternal death.
This sequence can occur within minutes, emphasizing that AFE is a true obstetric emergency demanding rapid recognition and aggressive support including oxygenation, hemodynamic stabilization, and management of coagulopathy.
How Does Amniotic Fluid Embolism Cause PPH?
Postpartum hemorrhage (PPH) in AFE results from severe coagulopathy, primarily due to Disseminated Intravascular Coagulation. The amniotic fluid contains tissue factor, which is a potent initiator of the extrinsic coagulation pathway. When this enters the maternal bloodstream, it leads to:
- Consumption of clotting factors and platelets
- Fibrin deposition in small vessels
- Subsequent hemorrhage due to depletion of coagulation components
In addition, uterine atony (loss of uterine muscle tone) often follows the hypoxic insult, worsening bleeding. This combination of consumptive coagulopathy and uterine atony makes AFE-induced PPH especially resistant to standard interventions such as uterotonics or surgical repair.
Management involves massive transfusion protocols, replacement of clotting factors, and supportive hemodynamic care.
Disseminated Intravascular Coagulation Causes and Risk Factors:
Disseminated Intravascular Coagulation (DIC) is one of the hallmark complications of AFE and is central to its high fatality rate. It occurs due to widespread activation of the coagulation cascade triggered by tissue factor and inflammatory mediators in the amniotic fluid.
Causes and Mechanisms:
- Activation of coagulation pathways by fetal antigens and tissue factor
- Endothelial damage from hypoxia and inflammatory mediators
- Consumption of platelets and fibrinogen, leading to uncontrolled bleeding
- Secondary fibrinolysis, further contributing to hemorrhage
Risk Factors for Developing AFE and DIC Include:
- Advanced maternal age (>35 years)
- Multiparity
- Placenta previa or abruption
- Cesarean or instrumental delivery
- Induction of labor
- Uterine rupture or trauma
These factors increase the likelihood of amniotic fluid entering the maternal bloodstream, predisposing patients to the immunologic storm that defines AFE.
Commonly Asked Questions about Amniotic Fluid Embolism Etiology (FAQs)
What is the pathophysiology of amniotic fluid embolism (AFE)?
The pathophysiology of AFE involves the entry of amniotic fluid components, including fetal tissue or meconium, into the maternal circulation. This can trigger a severe systemic inflammatory response syndrome, leading to hemodynamic instability, coagulopathy, and respiratory distress. The presence of amniotic fluid can initiate an anaphylactoid syndrome of pregnancy, further complicating the clinical scenario.
What are the common signs and symptoms of AFE?
The signs and symptoms of AFE typically occur suddenly during labor and delivery or immediately postpartum. Patients with AFE may present with acute respiratory distress, hypotension, altered mental status, and coagulopathy. Fetal heart rate abnormalities may also be observed, indicating fetal distress. Recognizing these signs and symptoms early is crucial for timely diagnosis and management.
What is the incidence of amniotic fluid embolism?
The incidence of AFE is relatively low, estimated to occur in about 1 in 15,000 to 1 in 30,000 deliveries. However, it is associated with a high maternal mortality rate, which has been reported to be as high as 60%. Understanding the risk factors for AFE can help in identifying at-risk patients during labor and delivery.
What are the risk factors for amniotic fluid embolism?
Risk factors for AFE include advanced maternal age, multiparity, cesarean delivery, and certain obstetric complications such as preeclampsia. Additionally, it may occur more frequently in patients with a history of trauma during labor or those undergoing rapid labor progression. Identifying these risk factors is vital for preventive measures.
How is the diagnosis of amniotic fluid embolism made?
Diagnosing amniotic fluid embolism is challenging and primarily based on clinical presentation. There is no definitive test for AFE, but clinicians often rely on a combination of symptoms, laboratory findings, and imaging studies. The presence of disseminated intravascular coagulation and the timing of symptom onset relative to labor and delivery can support the diagnosis.
What are the management options for amniotic fluid embolism?
The management of amniotic fluid embolism includes immediate resuscitation, fluid administration, and stabilization of the patient. Extracorporeal membrane oxygenation (ECMO) may be considered in severe cases to support respiratory and cardiac function. Early recognition and aggressive treatment are critical for improving outcomes and infant survival.
Are there any case reports of successful recovery from AFE?
Yes, there have been case reports documenting successful recovery from AFE, particularly with prompt intervention and supportive care. These cases often highlight the importance of early diagnosis and the implementation of advanced life support measures, demonstrating that there is a possibility for improved outcomes even in severe instances.
What is the mortality rate associated with amniotic fluid embolism?
The maternal mortality rate associated with AFE is notably high, with estimates ranging from 20% to 60%. This underscores the importance of understanding the clinical presentation and risk factors, as timely intervention can significantly impact maternal and fetal outcomes in cases of amniotic fluid embolism.
Conclusion
Amniotic Fluid Embolism is one of the most feared complications in obstetrics due to its sudden onset, complex pathophysiology, and high mortality. The evolving understanding of Amniotic Fluid Embolism Pathophysiology has shifted from a mechanical embolic concept to an immunologic and inflammatory model, emphasizing the role of maternal hypersensitivity and coagulopathy.
Early recognition, prompt resuscitation, and multidisciplinary management are critical for survival. Although prevention remains elusive, continuous research and improved critical care protocols are enhancing maternal outcomes. Postmortem investigations and detailed case analyses continue to deepen our understanding, bringing us closer to better diagnostic tools and lifesaving interventions.
In summary, pathophysiology of AFE serves as a reminder of the delicate balance between maternal and fetal systems during childbirth—and the importance of vigilance, preparedness, and swift action when this balance is disrupted.
Recommended posts
- How Common Is Amniotic Fluid Embolism: (Facts & Statistics)
- How to Avoid Amniotic Fluid Embolism? (Medical Guidance)
- How Does Amniotic Fluid Embolism Happen: (Causes & Risks)
- 9 Shocking Causes of Amniotic Fluid Embolism (Shouldn’t Ignore)
- Amniotic Embolism Survival Rate (The Latest Statistics)
- Amniotic Fluid Embolism Diagnosis (The Life-Saving Steps)
- Amniotic Fluid Embolism Incidence: How Rare Is It Really?
- Amniotic Fluid Embolism Risk Factors: A Comprehensive Guide
- The Deadly Link Between Amniotic Embolism and DIC Explained